
Overview
Mechanism of Action
Indications
Clinical Trials
Publications & Presentations
Overview
AC-1101 is a patented topical gel formulation whose active ingredient is a small-molecule Janus kinase (JAK) inhibitor, developed specifically for the treatment of autoimmune and inflammatory skin diseases.
Over the past decade, immunology and pathogenesis research has demonstrated the central role of the JAK–STAT signaling pathway in inflammation and autoimmune diseases. JAK inhibitors have been approved in many countries for the treatment of rheumatoid arthritis, psoriatic arthritis, ulcerative colitis, vitiligo, alopecia areata, and atopic dermatitis, showing broad clinical value.
The development of AC-1101 focuses on a patented formulation designed to enhance skin penetration and effectively modulate JAK–STAT signaling activated by multiple cytokines involved in skin diseases. Its potential applications include granuloma annulare, alopecia areata, atopic dermatitis, vitiligo, and other autoimmune skin disorders. As a topical therapy, AC-1101 also offers advantages for long-term use and may reduce the risk of systemic side effects associated with oral JAK inhibitors.


Mechanism of Action
Janus kinases (JAKs) are intracellular tyrosine kinases associated with type I and type II cytokine receptors. When cytokine binds to its receptor, JAKs are activated and undergo autophosphorylation, which in turn phosphorylates and activates STAT proteins. The activated STATs then form dimers and enter the nucleus to regulate the transcription of target genes, driving various inflammatory and immune-related responses.
By inhibiting JAK activity, AC-1101 effectively blocks this signaling cascade, preventing excessive STAT activation and modulating downstream inflammatory gene expression. This mechanism positions AC-1101 as a potential innovative therapy for autoimmune and inflammatory skin diseases.
1
Cytokine binds to its receptor
Signaling is initiated
2
JAK activation
Inflammatory signals are transmitted
3
STAT activation and translocation into the nucleus
Gene regulation
4
Downstream gene expression
Production of inflammatory mediators
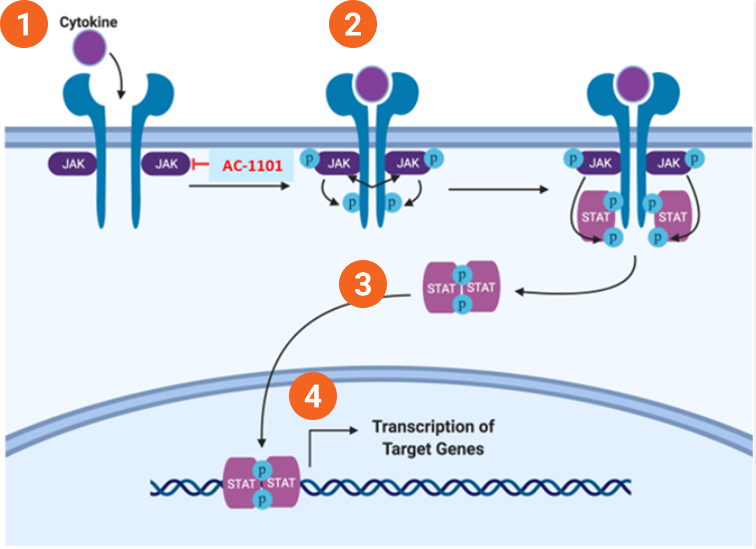
Note: Adapted from Yale J Biol Med. March 27, 2020; 93(1): 187–195.
Indications
Granuloma Annulare
Granuloma Annulare (GA) is a rare chronic dermatological condition characterized by red papules arranged in ring-shaped or arcuate lesions. Its prevalence in the United States is approximately 0.06–0.09%, and it occurs more frequently in women and middle-aged to older adults, with the highest incidence around the age of 50. Although large-scale epidemiological studies are lacking worldwide, clinical observations indicate that GA is not uncommon in general dermatology clinics.
The pathogenesis of GA is still under investigation. It is generally believed to be associated with an autoimmune response, in which chronic perivascular inflammation leads to alterations in dermal collagen, ultimately resulting in the characteristic annular skin lesions.
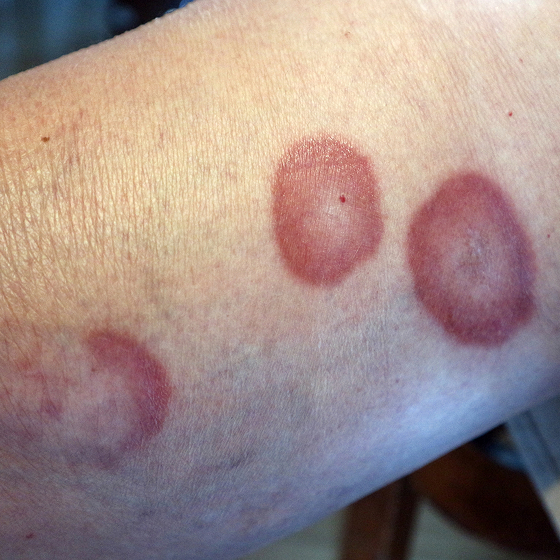

Clinical presentation of GA can be classified into five types:
Localized
The most common type of GA (approximately 75%), usually confined to the hands and feet.
Generalized
The second most common type (approximately 9.5–25%), with rashes spreading to the limbs and trunk.
Subcutaneous
Manifesteds as painless subcutaneous lumps or nodules, commonly found on the scalp, arms, and legs.
Perforating
Characterized by papules or pustules with yellow centers, which may exude fluid and form crusts, eventually leaving scars.
Patch-type
Appears as erythematous or brown patches with a smooth surface, lacking obvious papules or nodules. Despite the classification, GA patients may demonstrate more than one type of clinical presentation simultaneously.
Although GA is generally benign, its recurrent nature, prolonged course, and high relapse rate can cause significant cosmetic and psychological concerns for patients. Some patients have widespread or difficult-to-control lesions, requiring long-term medical visits and treatment. With no approved standard therapies for GA, clinical management mainly relies on corticosteroids, phototherapy, or immunosuppressants, which have limited efficacy and are prone to relapse. As a result, GA remains a condition with high unmet medical demand.
One Phase 1b clinical trial, sponsored by TWiB, to evaluate the efficacy and safety of topical AC-1101 gel in treating GA at Yale School of Medicine, led by Dr. William Damsky, was completed in 2024. Preliminary clinical results of this study show a favorable trend toward improved lesion appearance and inflammatory response, with good tolerability, which suggests that AC-1101 has the potential to become the first innovative treatment for GA, offering patients a viable therapeutic option.
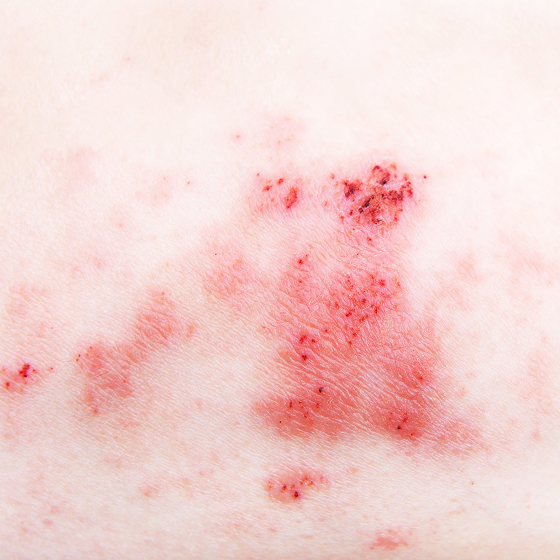

Atopic Dermatitis
Atopic dermatitis (AD) is one of the most common chronic inflammatory skin diseases in developed countries, affecting approximately 20% of children and 3% of adults. Globally, the estimated number of AD patients exceeds 200 million, including about 32 million in the United States, 30~40 million in Europe, and an estimated 1.5~2 million in Taiwan. Its prevalence continues to rise, posing a significant burden on public health and healthcare systems worldwide. Although most AD patients develop the disease in childhood, it often persists into adulthood and is characterized by recurrent flares and remissions.
Clinical manifestations include dry skin, chronic itching, erythematous rashes, edema, recurrent infections, and skin thickening with cracking (lichenification) due to prolonged scratching. These symptoms not only affect appearance but also significantly reduce quality of life, particularly impacting mental health and sleep. In children, AD can affect learning and social interactions, while caregivers face increased familial and psychological burdens.
Current treatments mainly rely on topical corticosteroids, immunosuppressants, and immunomodulators to reduce skin inflammation. However, these therapies often have long-term side effects or insufficient efficacy. Novel targeted therapies, such as JAK inhibitors, act on multiple pathogenic pathways and are considered highly promising treatment strategies, highlighting that atopic dermatitis continues to present substantial unmet medical needs in clinical practice.
Vitiligo
Vitiligo is a common acquired skin pigment disorder characterized by localized or widespread white patches. The global prevalence is estimated at approximately 0.5~2%, corresponding to roughly 40~80 million people worldwide. In the United States, the prevalence is around 0.76%, translating to about 2.5 million patients; European reports typically range from 0.5~1.0%; and data from Taiwan’s National Health Insurance database indicate a prevalence of approximately 0.4~0.6%, or an estimated 100,000~150,000 people.
Vitiligo can occur at any age, but onset is most common in adolescence or young adulthood. Clinical subtypes include localized, segmental, and generalized forms. Although vitiligo itself rarely causes physical pain or life-threatening risk, changes in skin appearance often lead to significant psychological stress, social impact, and reduced quality of life.
Current treatments primarily involve topical therapies (such as corticosteroids and calcineurin inhibitors) and phototherapy, but these approaches have limited efficacy and high relapse rates. Emerging research indicates that JAK inhibitors (JAKi) can act on the IFN-γ / JAK / STAT pathway, suppress pathogenic signals such as CXCL10, and block the immune mechanisms that destroy melanocytes. This positions JAKi as one of the most promising innovative therapies for vitiligo.




Alopecia Areata
Alopecia Areata is an autoimmune disease caused by the immune system abnormally attacking hair follicles, resulting in non-scarring hair loss. It affects approximately 2% of the general population worldwide over their lifetime. In the United States, the prevalence is approximately 0.1–0.2%, affecting an estimated 300,000 to 600,000 individuals.
The clinical presentation of AA varies significantly in severity. It most commonly manifests as patchy alopecia, presenting as smooth, round, or oval bald patches on the scalp or beard area. However, the disease can progress to more severe forms, such as Alopecia Totalis (complete loss of scalp hair) or Alopecia Universalis (complete loss of hair on the scalp and body). While AA is not life-threatening, the unpredictable nature of hair loss and the visible alteration of appearance often lead to profound psychological distress, anxiety, and depression, severely impacting the patient's quality of life.
The pathogenesis of AA is driven by an autoimmune attack on the hair follicle, resulting in the collapse of the follicle's "immune privilege." Research has identified that this process is dependent on the JAK–STAT signaling pathway, specifically involving cytokines such as IFN-γ that recruit CD8+ T cells to attack the hair follicle.
Current standard-of-care treatments include intralesional corticosteroids and, more recently, oral JAK inhibitors. While oral JAK inhibitors have shown efficacy, the long-term use of oral JAK inhibitors carries potential safety concerns, requiring continuous monitoring for risks such as infections, cardiovascular events, and other systemic side effects. Currently, no new topical formulations have been approved for Alopecia Areata. Consequently, there remains a significant unmet clinical and market demand for a topical formulation that offers a safe and effective option for long-term use.
AC-1101 as a topical formulation, aims to address this unmet need by delivering high concentrations of the JAK inhibitor directly to the hair follicle site. This targeted approach is designed to effectively block the inflammatory signaling loops responsible for hair loss while minimizing systemic exposure and safety concerns.
Clinical Trials
- April 2020: An Investigational New Drug (IND) application to conduct clinical trials of AC-1101 for the treatment of vitiligo was approved by the US FDA.
- November 2020: Completed a Phase I clinical trial on AC-1101 in healthy volunteers to evaluate safety and pharmacokinetics.
- July 2022: Approved by the US FDA for an IND application to conduct clinical trials of AC-1101 for the treatment of granuloma annulare.
- November 2022: Initiated a Phase Ib clinical trial in GA patients in the United States.
- July 2024: The US Phase Ib trial of AC-1101 for GA was completed, meeting its objectives and demonstrating favorable safety, tolerability, and PK profiles, alongside a preliminary therapeutic trend.
ClinicalTrials.gov Identifier:
-
Phase I clinical trial on safety and pharmacokinetics in healthy subjects (AC-1101-PK-001): NCT04468425(https://clinicaltrials.gov/ct2/show/NCT04468425)
-
Phase 1b Trial of Granuloma Annulare (AC-1101-GA-001): NCT05580042 (https://clinicaltrials.gov/ct2/show/NCT05580042)
Publications & Presentations
•Wang A, Rahman NT, McGeary MK, Murphy M, McHenry A, Peterson D, Bosenberg M, Flavell RA, King B, Damsky W. Treatment of granuloma annulare and suppression of proinflammatory cytokine activity with tofacitinib. J Allergy Clin Immunol. 2021 May;147(5):1795
•Wang A, Singh K, Ibrahim W, King B, Damsky W. The Promise of JAK Inhibitors for Treatment of Sarcoidosis and Other Inflammatory Disorders with Macrophage Activation: A Review of the Literature. Yale J Biol Med. 2020 Mar 27;93(1):187-195


